If you’ve made it this far then I’m willing to bet Christmas is almost upon us...
but there might just be enough time left to make one extra ornament for the Christmas tree...
and learn a little bit about how crystals are formed too!
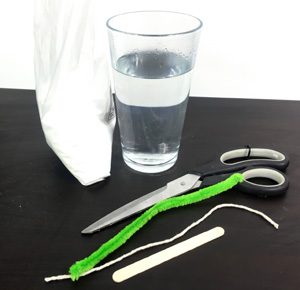
What Do I Need?
- Borax
- Pint of water
- Pipe cleaner
- Scissors
- Stick
- Sting

How Do I Do It?
STEP1 - To make sure our Crystal Canes don’t get stuck to the side of the glass the first step is to get an adult to help cut our pipe cleaner in half.
WARNING: Never drink Borax. Make sure you label and store it correctly and wash your hands after your finish experimenting!
STEP2 - Get an adult to help out with filling your pint glass with some very warm water.
STEP3 - Add a table spoon of borax at a time and stir it in. Keep going until you can’t dissolve anymore and there is a white layer at the bottom of the glass.
STEP4 - Tie a piece of string around your pipe cleaner cane and your stick.
STEP5 - Lower our pipe cleaner cane into you glass making sure it doesn’t touch the bottom or the sides.
STEP6 - Leave overnight for your Crystal Cane to form!

What’s Going On?
Alright, alright! The eagle-eyed amongst you will have noticed that this experiment is pretty similar to the Scientific Snowflake.
Who doesn’t want an extra ornament for their Christmas tree as well as an awesome way to reinforce our learnings about crystallisation.
When you mix your borax with your warm water so that some remains undissolved you maximised the amount of borax that the water can hold.
As the water begins to cool the amount of borax your water can ‘hold’ starts to come down. Your borax is ‘looking’ for somewhere to go.
Your pipe cleaner cane is kind of rough and offers the perfect place for your borax to settle (as opposed to the smooth sides of your glass).

More Fun Please! - Experiment Like A Real Scientist!
- How does the temperature of the water affect the outcome of this experiment?!
Want A FREE Science Experiment Book?
If you like fun science experiments then you'll love this FREE Science experiment book....
It's packed full of awesome experiments you can do at home with "stuff" you've already got.
Your FREE Science experiment book is:
- 5 Star Reviewed
- Dragons' Den Approved
- Yours completely FREE.
Kids Party Coming Up?
If you like fun science experiments then you’ll love the Dragons’ Den winning Sublime Science Party. Check price & availability in your area and grab your FREE Kids Party Survival Guide right now!

Home - About - Privacy Policy - Terms & Conditions - Facebook - Instagram - LinkedIn - Twitter - Sitemap - Blog - Contact
Copyright: Sublime Science Labs Ltd - All Rights Reserved - Registered in England and Wales, Company Number 15933481
Registered Address: Unit 7 Global Business Park, 14 Wilkinson Rd, Cirencester GL7 1YZ - Call: 0116 380 0750


